 
Like other chemistry activities, these valence electron activities are the perfect way to make an abstract concept more engaging, especially for kinesthetic learners. Thus, as size (atomic radius) increases, the ionization energy should decrease. This easy way to teach about valence electrons will help students connect the concept of electrons to the periodic table. Remember, shells don’t neatly stack on top of each other, so valence (and oxidation state. The most stable oxidation state is one that fills or half-fills an atom’s electron shell. Usually it’s oxidation state is +1, but it can lose the electron and have a valence of -1. 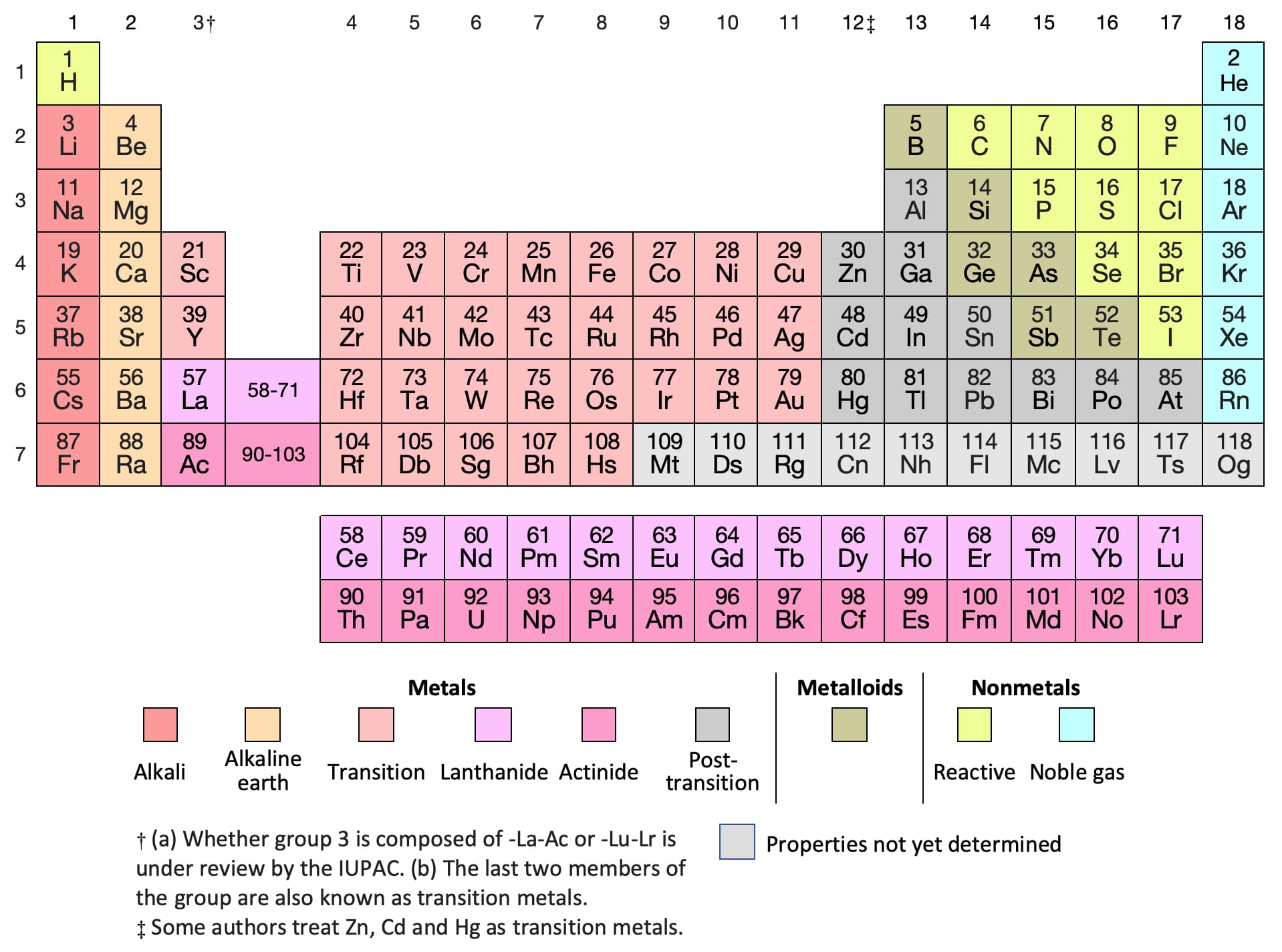
For larger atoms, the most loosely bound electron is located farthest from the nucleus and so is easier to remove. A lithium atom has one outer shell electron. Energy is always required to remove electrons from atoms or ions, so ionization processes are endothermic and IE values are always positive. The energy required to remove the third electron is the third ionization energy, and so on. This activity can stand alone or be used with the attached worksheet and/or create other worksheets when discussing atom radius or ion radius.\] Because much of the chemistry of an element is influenced by valence electrons, we would expect that these elements would have similar chemistryand they do. It helps them learn it better than reading it off another chart and it gives them ownership so they keep them longer. They all have a similar electron configuration in their valence shells: a single s electron. The number of valence electrons depends on the octet rule. Elements within the same group share the same number of valence electrons. Atoms in a period have the same number of electron shells. Lithium has a single electron in the second principal. An element period is a horizontal row on the periodic table. In the second period elements, the two electrons in the 1 s sublevel are called inner-shell electrons and are not involved directly in the elements reactivity, or in the formation of compounds. 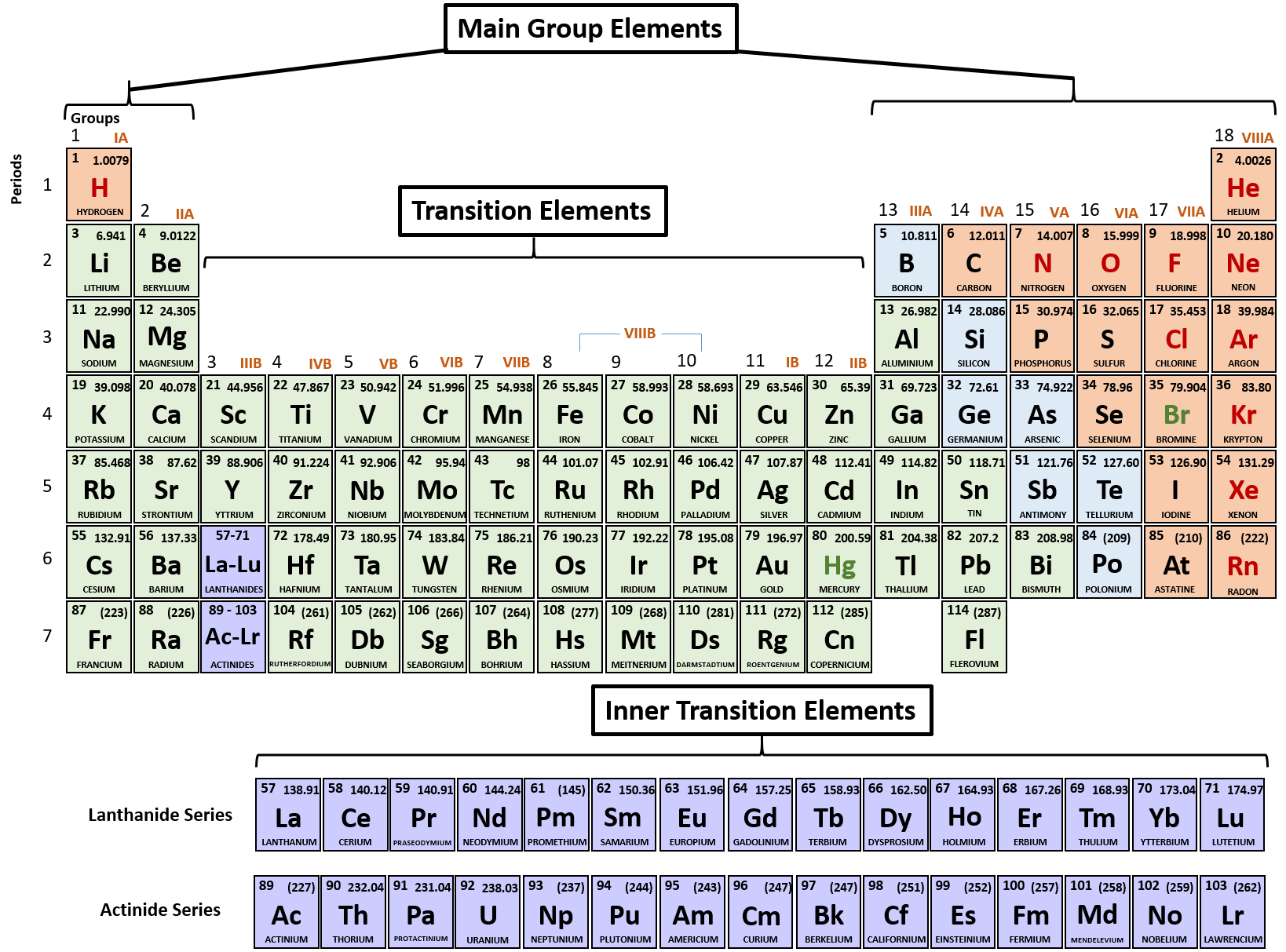
Have students add information to the back of their tables like polyatomic ions, equations, key concepts, exceptions to rules. Valence electrons are the electrons in the highest occupied principal energy level of an atom. Have students reference their tables when introducing concepts like atomic and ionic radius trends, ionic and covalent bonding regions. 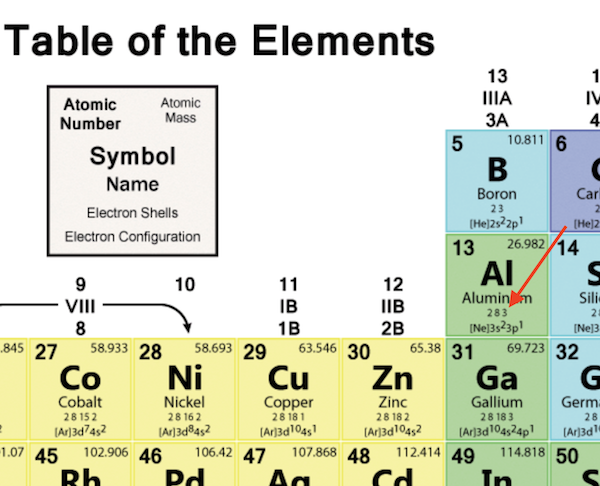
It is the transition metal due to which it has the variable valency. The valency of gold may be either of 1 or 3 in different scenarios. The interaction of valence electrons helps in understanding the chemical binding of elements. This works best when either all the information is on the table or when discussing properties. It will further provide the accurate numbers of valence electrons of atoms. Information that could be included, but is not limited to, are element name, atomic numbers, mass numbers or weights, common oxidation numbers.Īt some point the columns could be lightly shaded and a legend could be added. This activity has students place the electron-dot model in each appropriate box, but if boxes are large enough other information maybe added throughout the year. The valence is the combining capacity of an atom of a given element, determined by the number of hydrogen atoms that it combines with. Make sure students are filling in the boxes correctly from the start! We can see from the electron configuration of a carbon atom1 s2 2 s2 2 p2 that it has 4 valence electrons (2 s2 2 p2) and 2 core electrons (1 s2 ). Have students keep their tables for a reference tool. Valence shell electrons (or, more simply, the valence electrons) are the electrons in the highest-numbered shell, or valence shell, while core electrons are the electrons in lower-numbered shells. The valence electrons are symbolized as a dot and placed in increasing number around the symbol.Įven though the numbering scheme is no longer widely used it will be helpful later when introducing the idea of half-filled S and P sub-orbital's. Where X is the symbol of the element and accounts for the nucleus and all the filled inner shell electrons. It is very helpful to model a couple of electron-dot examples to get students started. Adding the atomic numbers in the lower corner allows for the addition of mass numbers or atomic weights later if desired. How many valence electrons does an element have You can use the periodic table to help you determine how many valence electrons an element (specifically, a neutral atom of the element) has. It maybe helpful to have students number the boxes one through twenty (atomic numbers) in the lower left corner first to avoid confusion. On the other hand, nitrogen can form NH 3 so it has a valence of 3, and 3 valence electrons. In this short activity (20-25 minutes) students will be provided a blank periodic table and will fill in the electron-dot model for the first twenty elements. This activity allows students to make a tool of models to help them visualize concepts of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
